Aqa periodic table: GCSE Chemistry Revision Guide (Groups, Periods & Atomic Structure Explained)
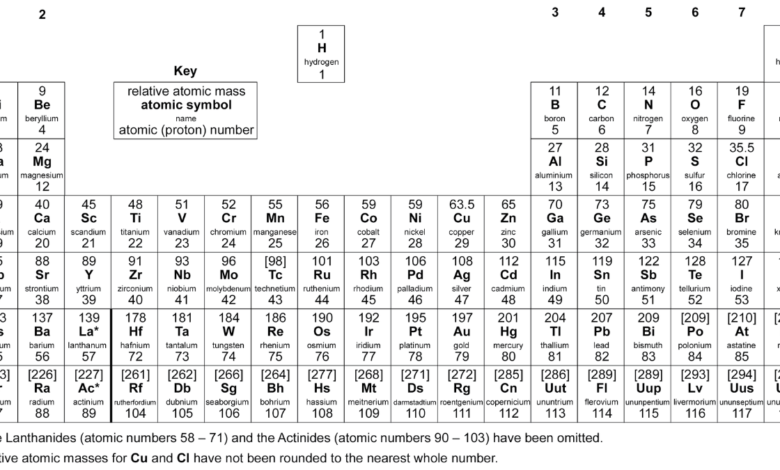
The aqa periodic table is one of the most important tools used in GCSE and A-Level Chemistry across the UK education system. It organises all known chemical elements in a structured format based on atomic number, allowing students to understand patterns in chemical behaviour. By learning how elements are arranged, learners can better interpret reactions, trends, and scientific relationships.
In modern science education, the periodic table of elements is essential for building a strong foundation in chemistry. The aqa periodic table is specifically designed to support students in exams by highlighting key groups and trends. It simplifies complex scientific ideas, making it easier for learners to understand atomic structure, chemical properties, and how elements interact in real-world situations.
Structure and Organisation of the Periodic Table
The structure of the aqa periodic table is based on increasing atomic number, which represents the number of protons in each atom. Elements are arranged in rows called periods and columns known as groups. This organisation helps students identify patterns, such as how elements behave similarly when they are in the same group.
Understanding the structure is essential in both GCSE and A-Level Chemistry. The periodic table gcse specification focuses on basic organisation, while advanced learners at A-Level explore deeper scientific explanations. The aqa periodic table helps students recognise how electron configuration influences chemical properties and why elements in the same group react in similar ways.
AQA Periodic Table for GCSE Chemistry Students
The aqa periodic table plays a key role in GCSE Chemistry revision, especially when studying Groups 1, 7, and 0. Group 1 elements, known as alkali metals, become more reactive as you move down the group. Group 7 elements, called halogens, show decreasing reactivity, while Group 0 noble gases remain stable due to full outer electron shells.
For GCSE students, the aqa gcse periodic table is used in almost every topic involving chemical reactions and atomic structure. It helps learners answer exam questions about trends, properties, and element identification. By understanding the periodic table aqa format, students can improve their confidence and accuracy in both structured and multiple-choice questions.
Advanced Understanding in A-Level Chemistry
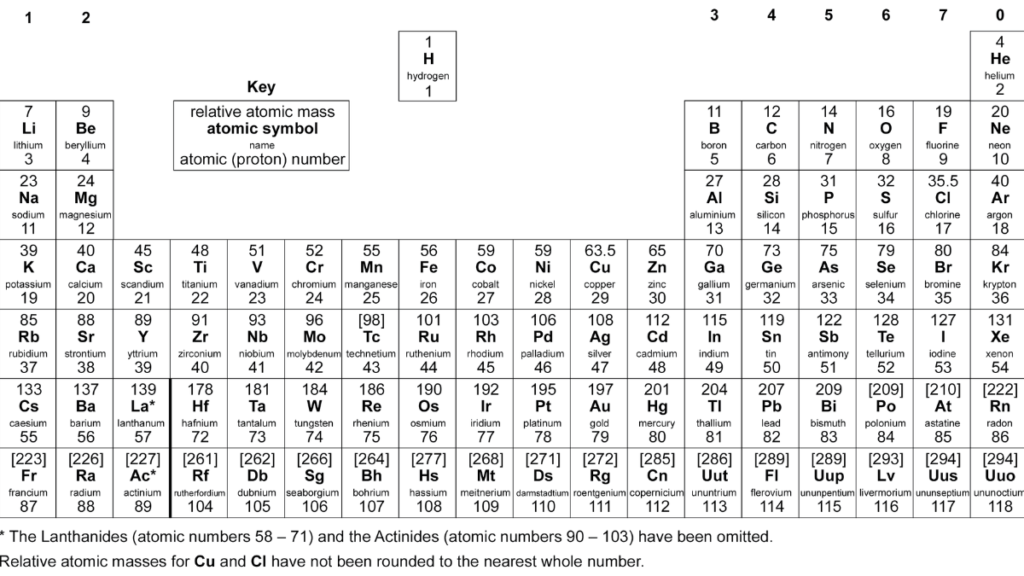
At A-Level, the aqa periodic table becomes more detailed and analytical. Students are expected to understand why trends occur, not just memorise them. Topics such as ionisation energy, atomic radius, and electron shielding are introduced, providing a deeper explanation of periodic behaviour across elements.
The aqa a level periodic table also includes transition metals and complex ions, which require a more advanced understanding of chemical bonding and reactions. Compared to GCSE, the a level periodic table chemistry syllabus focuses on data interpretation and scientific reasoning, helping students develop higher-level analytical skills needed for university studies.
Historical Development of the Periodic Table
The periodic table of elements has a rich scientific history that dates back to 1869. It was first successfully developed by Dmitri Mendeleev, a Russian scientist who organised elements by atomic mass and predicted the existence of undiscovered elements. His work laid the foundation for the modern periodic table used today.
Over time, scientists refined Mendeleev’s system, leading to the modern structure used in the aqa periodic table. Today, elements are arranged by atomic number instead of atomic mass. This improvement made the periodic table more accurate and reliable, allowing chemists to better understand the relationships between elements.
Exam Techniques for the AQA Periodic Table
When studying the aqa periodic table, exam technique is just as important as knowledge. Students are often asked to explain trends such as reactivity, atomic size, and group similarities. Clear scientific explanations supported by periodic trends are essential for achieving high marks in both GCSE and A-Level exams.
Practising questions based on the gcse periodic table helps students become familiar with common exam patterns. In A-Level Chemistry, the aqa periodic table is used in more complex questions involving calculations and data analysis. Understanding how to interpret the table quickly can significantly improve exam performance and reduce errors.
Comparison of Exam Board Periodic Tables
Different UK exam boards use similar periodic table structures, but there are slight differences in focus and detail. The ocr periodic table and the aqa periodic table follow the same scientific principles, but their specifications may vary in terms of depth and emphasis on certain topics.
Students studying the periodic table ocr a level or aqa a level chemistry periodic table should focus on understanding core scientific principles rather than memorising differences. The underlying structure of the periodic table remains consistent, making it easier for students to adapt across different exam boards.
Real-World Applications of the Periodic Table
The aqa periodic table is not only important for exams but also for understanding real-world chemistry. Scientists use it to develop new medicines, design materials, and create chemical solutions for industrial and environmental challenges. It plays a vital role in modern scientific innovation.
From healthcare to engineering, the periodic table of elements helps researchers predict how substances will behave. The periodic table aqa system allows students to connect classroom learning with real-life applications, making chemistry more meaningful and practical beyond academic study.
Importance of Learning Element Names and Symbols
Understanding the periodic table with names and symbols is essential for success in chemistry. Each element has a unique symbol, such as H for hydrogen and O for oxygen, which is used in chemical equations and scientific communication. The aqa periodic table helps students learn these quickly and accurately.
In both gcse periodic table and a level chemistry studies, recognising element names is crucial for interpreting reactions and solving problems. The periodic table aqa format ensures students become familiar with key elements, improving their ability to answer exam questions confidently and correctly.
Conclusion
The aqa periodic table is a fundamental part of GCSE and A-Level Chemistry, providing a structured way to understand all known elements. It helps students identify patterns, learn chemical properties, and build a strong foundation in scientific knowledge required for exams and beyond.
By mastering the periodic table gcse and advancing to a level chemistry periodic table concepts, students can develop essential analytical skills. The aqa periodic table not only supports academic success but also prepares learners for real-world scientific understanding and future studies in chemistry.
Frequently Asked Questions
What is the AQA periodic table used for?
It is used in GCSE and A-Level Chemistry to organise elements and study their properties and trends.
Who created the original periodic table?
Dmitri Mendeleev, a Russian scientist, created the first successful periodic table in 1869.
What is the difference between GCSE and A-Level periodic table knowledge?
GCSE focuses on basic trends, while A-Level includes deeper scientific explanations and calculations.
Why is the periodic table important in chemistry?
It helps explain how elements behave and how they react with each other in chemical processes.
What are the main groups in the periodic table?
Group 1 alkali metals, Group 7 halogens, and Group 0 noble gases are the most commonly studied groups.
Also Read: Faversham



